- In 2014, the Royal Swedish Academy of Sciences proclaimed that “the 21st century will be lit by LED lamps,” recognising the importance of Light-Emitting Diodes (LEDs) in influencing the future of lighting technology.
- This article goes into the interesting world of diodes, LEDs, and their role in modern technology.
Understanding diodes
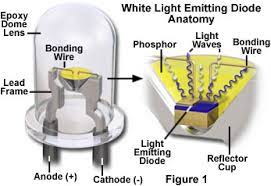
- Diode Basics: A diode is a tiny electrical component having two terminals: anode and cathode. Its principal function is to enable current to flow in just one way via a p-n junction.
- A p-n junction is made up of two neighbouring materials: a p-type with positive charge carriers known as holes, and an n-type with negative charge carriers known as electrons. Electrons may readily flow from the n-type to the p-type but not the other way around, allowing the diode to regulate current in one direction alone.
- Anode and Cathode: The anode terminal is linked to the p-type material, while the cathode is connected to the n-type material. These terminals determine the diode’s directionality.
The birth of light-emitting diodes (LEDs)
- Electroluminescence: LEDs are light-emitting diodes. Electrons, which have greater energy levels than holes, release energy when they occupy holes in the p-n junction. If this energy falls inside the visible spectrum, it emits light, a process known as electroluminescence.
- LEDs create certain light colours by ensuring that electron-hole recombination produces a correct quantity of energy, which is determined by the band gap.
The Band Gap’s significance
- It lies in the fact that electrons can only have specific energy levels. These electrons typically occupy the lowest energy levels accessible, resulting in conductors, insulators, and materials with a band gap.
- Band Gap’s Role: A band gap denotes the energy threshold necessary for electrons to transition from lower to higher energy levels, allowing materials to conduct electricity.
- LED and Band Gap: The energy emitted during electron-hole recombination in LEDs correlates to the band gap, which determines the light’s colour.
The colour palette of LEDs
- It has evolved over four decades, with red and green LEDs coming first. The objective was to create gallium nitride crystals with precise electroluminescent characteristics.
- Primary Colours: LEDs may emit red, green, and blue light, providing a diverse colour pallet. Combining multiple LEDs results in a wide range of colours on display boards and displays.
- Breakthrough: In the late 1980s, Japanese researchers Isamu Akasaki, Hiroshi Amano, and Shuji Nakamura achieved a big breakthrough by producing a brilliant blue LED out of gallium nitride. Their achievements earned them the 2014 Nobel Prize in Physics.
Advantages of LEDs
- LEDs are more efficient than incandescent bulbs and fluorescent lights, producing more light per watt of electricity. This is one of its advantages.
- LEDs are extremely durable, resulting in less material waste and lower maintenance costs.
- LEDs are used in a variety of applications, including consumer electronics and signs, greenhouse illumination, and air quality monitoring.
- Colour versatility: LEDs can emit a variety of colours and frequencies, making them suitable for a wide range of applications.
Future Prospects
- Haitz’s Law: Like Moore’s law, LEDs have followed Haitz’s law, which predicts cost reduction and greater light output over time.
- Innovations: Ongoing research investigates skin-embedded LEDs, organic LEDs, and efficient LEDs produced from perovskites, all of which promise significant breakthroughs in lighting technology.
Source: https://www.sciencedirect.com/topics/engineering/light-emitting-diode