Over other forms of treatment, the new CAR T-Cell Immunotherapy holds promise for Ovarian Cancer patients.
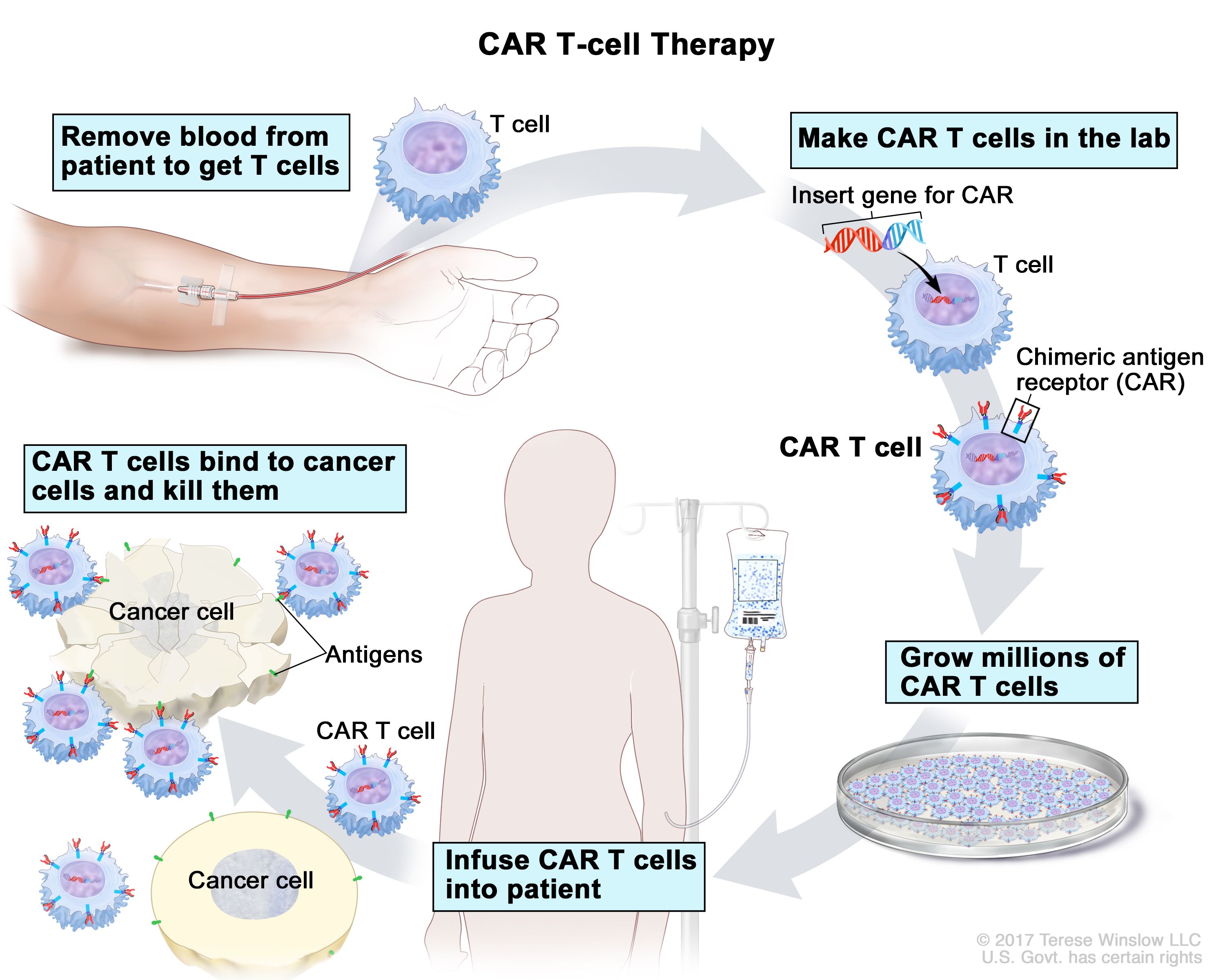
What are CAR T-cells?
- Chimeric antigen receptor (CAR) T-cell therapies represent a quantum leap in cancer treatment sophistication.
- CAR T-cell therapies use a patient’s own cells, as opposed to chemotherapy or immunotherapy, which require mass-produced injectable or oral medication.
- They are genetically modified in the lab to activate T-cells, a type of immune cell, to attack tumours.
- After conditioning the cells to multiply more effectively, they are infused back into the patient’s bloodstream.
- The cells are more specific than targeted agents and directly activate the patient’s immune system against cancer, increasing clinical efficacy.
- This is why they’re called ‘living drugs’.
How does the therapy function?
- CAR T-cell therapy involves drawing the patient’s blood to harvest T-cells, which are immune cells that play an important role in destroying tumour cells.
- In the laboratory, researchers modify these cells so that they express specific proteins on their surfaces, known as chimeric antigen receptors (CAR).
- They have a preference for proteins found on the surface of tumour cells.
- This change in cellular structure allows CAR T-cells to effectively bind to and destroy tumours.
- The patient’s immune system clears the tumour as the final step in its destruction.
Where is it used?
- As of today, CAR T-cell therapy has been approved for leukaemias (cancers arising from the cells that produce white blood cells) and lymphomas (arising from the lymphatic system).
- These cancers are caused by the uncontrolled reproduction of a single clone of cells, which means that after a single type of cell undergoes cancerous transformation, it produces millions of identical copies.
- As a result, the CAR T-cell target is consistent and reliable.
- CAR T-cell therapy is also used in patients whose cancers have returned after a successful initial treatment or who have not responded to previous chemotherapy or immunotherapy combinations.
- Its response rate varies. The efficacy in certain types of leukaemias and lymphomas can reach 90%, whereas it is significantly lower in other types of cancer.
How widespread is its application?
- The difficulty in preparing CAR T-cells has been a major impediment to their use.
- The first clinical trial demonstrating their efficacy was published nearly a decade ago, but the first indigenously developed therapy in India was only successfully performed in 2022.
- This therapy necessitates a significant amount of technical and human resources.
- Treatments in the United States can cost more than a million dollars.
- Trials are currently underway in India, with companies attempting to manufacture CAR T-cells at a fraction of the cost.
- The preliminary findings are encouraging.
What exactly are conventional cancer treatments?
- Surgery (removing the cancer), radiotherapy (delivering ionising radiation to the tumour), and systemic therapy are the three main types of cancer treatment (chemotherapy- administering medicines that act on the tumour only).
- Surgery and radiotherapy have been greatly refined over time, while advances in systemic therapy have been unprecedented.
- CAR T-cell therapy is a new development in this field that is capturing the attention of many researchers worldwide.
Will this therapy be as expensive in India?
- Any new therapy introduced in India faces the twin challenges of cost and value.
- Critics argue that developing facilities in India may be redundant and/or inappropriate because CAR T-cell therapy will be unaffordable to the majority of Indians even if it becomes cheaper.
- Those who can afford it and need it are already receiving it abroad.
- While this is correct, it may be the correct answer to the incorrect question.
- Every patient has the right to access a global standard of care; the next step is to figure out how to make it more affordable.
Source: https://www.cancer.gov/about-cancer/treatment/research/car-t-cells
